X-Pure® : a range of purified modified and non-modified gelatins for research and in body applications
Designed by Nature, Perfected by Rousselot
X-Pure® is a range of pharmaceutical-grade, customizable, low endotoxin gelatins suitable for a wide range of biomedical applications, from in vitro diagnostic medical devices to in body commercial products. The range includes:
Rousselot’s X-Pure range has been developed to ensure maximum quality and safety. X-Pure gelatins and GelMAs are manufactured to a GMP standard*, including quality and regulatory compliance. Our research and GMP grades are functionally equivalent, minimizing the need to revalidate data ahead of clinical trials and helping to accelerate research to clinic translation.
The Advantages of X-Pure
Ultra-low endotoxin gelatins for maximum quality & safety
Endotoxins can impact cell viability and proliferation and cause immune reactions in patients. In order to be accepted onto the market, biomedical products and medical devices must comply with strict endotoxin limits imposed by the FDA and other regulatory bodies.
All X-Pure gelatins support regulatory compliance with international guidelines. With endotoxin levels of (<10 endotoxin units (EU)/g), they are amongst the lowest available on the market – over 100 times lower than traditionally manufactured gelatins. Our patented process** removes Lipopolysaccharide (LPS) endotoxins and process impurities.
Watch our video: “Bridging the valley of death” to find out how choosing biomaterials with ultra-low endotoxins can help to bridge the clinical translation gap.
Tunable characteristics with guaranteed batch-to-batch consistency
X-Pure gelatins have tunable molecular weight and can be chemically functionalized to meet your specific needs. Our modified gelatins such as GelMA and GelDAT offer stability at body temperature, coupled with tunable gel strength and degree of functionalization. Our process delivers batch-to-batch consistency and purity, for reliable results every time.
Full traceability, readily available documentation & validated viral safety
X-Pure gelatins come with full traceability, readily available documentation and validated viral safety, helping to save time when reaching the clinic.
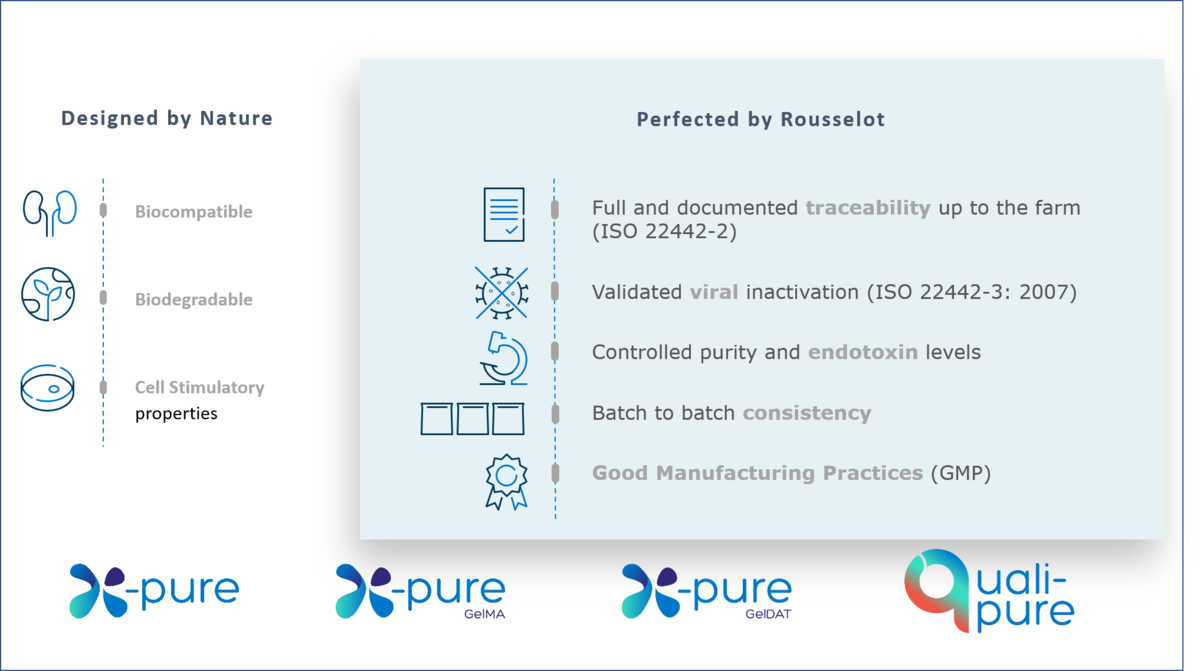
Medical device producers are facing increased regulatory requirements for biomaterials. From May 2024, all medical devices placed on the European market must conform with the new EU Medical Device Regulation (MDR). Regulation (EU) 2017/745 on Medical Devices (the MDR) has replaced the existing medical devices Directive (93/42/EEC) (MDD) and the active implantable medical devices Directive (90/385/EEC) (AIMDD). In addition, the ISO standard 22442 has been updated to bring it further in line with global requirements for medical devices. X-Pure provides the supporting documentation for full compliance of medical devices with the new MDR requirements and with ISO 22442.
Get in touch or check out our Product Finder to find the ideal biomaterial for your application.
* IPEC & PQG Joint Good Manufacturing Practices Guide for Pharmaceutical Excipients
**Patent WO2016085345
![[Translate to Chinese:] X Pure range [Translate to Chinese:] Woman looking at the samples](https://d1ip4j1950xau.cloudfront.net/_processed_/2/b/csm_woman-choosing-products-pharmacy-ConvertImage_d4ed8b14a1.jpg)


